Molecular and Cellular Neurosciences
- Molecular and Cellular Neurosciences (Laboratory of Molecular Synaptic Function)
- Molecular and Cellular Neurosciences (Laboratory of Neural Membrane Biology)
- Molecular and Cellular Neurosciences (Laboratory of Developmental Neurobiology)
Laboratory of Developmental Neurobiology

Jun Motoyama, Ph.D.
We believe that understanding the developmental process of the brain will lead us to understanding of the brain, so we are focusing on two themes.
The first theme is how neural stem cells develop. Neural stem cells proliferate by themselves early embryonic stage and increase in the number of stem cells, but eventually they begin to produce neurons and, over time, glial cells. We're looking at changes in neural stem cells over time. Why does the nature of stem cells change over time? To date, we have observed spontaneous changes in intracellular Ca2 + concentration in neural stem cells. We found that this variation pattern (Period and Width) was different in the early and late stages of neural stem cell development. Changes in the pattern of spontaneous changes in intracellular Ca2 + concentration may be related to changes in stem cell properties that occur over time. This phenomenon may lead us to understand the molecular mechanisms involved in the development of neural stem cells.
The second theme is how the brain develops after birth. It is known that mother and child separation stress affect the development of the hippocampus and the function of the brain after growth. The brain develops under the influence of the environment, but the detailed mechanism is not well understood. Using a system of maternal separation stress, we found that the differentiation of inhibitory neurons was suppressed by this stress, and that the sensitivity to stress differed between the left and right hippocampus. With this phenomenon as an opportunity, we are trying to clarify the relationship between environmental factors and the differentiation control mechanism of inhibitory nerve cells in the brain development process.
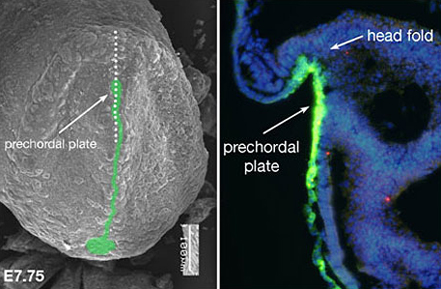
Fig.1 : Expression of Sonic hedgehog (Shh) protein and the determination of the midline structure in mouse embryo
An SEM micrograph of the frontal view of a mouse embryo (fetal age 7.75 days) Left: Shh protein is green. Right: Enlarged, cross-section of an embryo at the same developmental day. The dotted line in the micrograph (left) shows the region: Shh antibody reveals Shh. The part that will become the brain (head fold) is followed by the perchordal plate. Shh (in green) that is expressed in the prechordal plate induces midline structure formation.
Research theme
- Understanding the Molecular Mechanisms that Control Neural Stem Cell Development.
- Effect of Maternal Separation Stress on Hippocampal Development in Neonates.
- Spontaneous fluctuation of intracellular Ca2 + concentration during mammalian neural stem cell development.
Selected publications
- Murakami Y, Imamura Y, Saito K, Sakai D, Motoyama J. (2019) Altered kynurenine pathway metabolites in a mouse model of human attention-deficit hyperactivity/autism spectrum disorders: A potential new biological diagnostic marker. Sci Rep. Sep 12;9(1):13182.
- Watanabe-Takahashi M, Yamasaki S, Murata M, Kano F, Motoyama J, Yamate J, Omi J, Sato W, Ukai H, Shimasaki K, Ikegawa M, Tamura-Nakano M, Yanoshita R, Nishino Y, Miyazawa A, Natori Y, Toyama-Sorimachi N, Nishikawa K. (2018) Exosome-associated Shiga toxin 2 is released from cells and causes severe toxicity in mice. Sci Rep. Jul 17;8(1):10776.
- Katahira T, Miyazaki N, Motoyama J. (2018) Immediate effects of maternal separation on the development of interneurons derived from medial ganglionic eminence in the neonatal mouse hippocampus. Dev Growth Differ. Jun;60(5):278-290.
- Shikata Y, Okada T, Hashimoto M, Ellis T, Matsumaru D, Shiroishi T, Ogawa M, Wainwright B, Motoyama J. (2011) Ptch1-mediated dosage-dependent action of Shh signaling regulates neural progenitor development at late gestational stages. Dev Biol. Jan 15;349(2):147-159.
- Aoto K., Shikata Y., Imai H., Matsumaru D., Tokunaga T., Shioda S., Yamada G. & Motoyama J. (2009) Mouse Shh is required for prechordal plate maintenance during brain and craniofacial morphogenesis. Dev Biol. 327, 106-120.
- Aoto K., Shikata Y., Higashiyama D., Shiota K. & Motoyama J. (2008) Fetal ethanol exposure activates protein kinase A and impairs Shh expression in prechordal mesendoderm cells in the pathogenesis of holoprosencephaly. Birth Defects Res A Clin Mol Teratol. 82, 224-231.
- Motoyama J. (2006) Essential roles of Gli3 and sonic hedgehog in pattern formation and developmental anomalies caused by their dysfunction. Congenit Anom. 46, 123-128. Review.
- Motoyama J. & Aoto K. Important role of Shh controlling Gli3 functions during the dorsal-ventral patterning of the telencephalon. In Hedgehog-Gli Signaling in Human Disease, Editor: A. Ruiz i Altaba Chapter 14, 177-183 (2006). Landes Bioscience/Eurekah.com, Springer Science+Business Media, Inc. Texas. Review.
- Motoyama J., Milenkovic L., Iwama M., Shikata Y., Scott M.P., & Hui CC. (2003) Differential requirement for Gli2 and Gli3 in ventral neural cell fate specification. Dev Biol. 259, 150-161.
- Aoto K., Nishimura T., Eto K. & Motoyama J. (2002) Mouse GLI3 regulates Fgf8 expression and apoptosis in the developing neural tube, face, and limb bud. Dev Biol. 251, 320-332.
- Motoyama J., Liu J., Mo R., Ding Q., Post M. & Hui CC. (1998) Essential function of Gli2 and Gli3 in the formation of lung, trachea and oesophagus. Nat Genet. 20, 54-57.
- Motoyama J., Takabatake T., Takeshima K. & Hui CC. (1998) Ptch2, a second mouse Patched gene is co-expressed with Sonic hedgehog. Nat Genet. 18, 104-106.
- Motoyama J., Kitajima K., Kojima M., Kondo S. & Takeuchi T. (1997) Organogenesis of the liver, thymus and spleen is affected in jumonji mutant mice. Mech Dev. 66, 27-37.
Members
Jun Motoyama, Ph.D. (Principal investigator, Professor)
Yuki Ito, Ph.D.(Research assistant professor)
Yoko Shimada, Ph.D. (Research assistant professor)
Contact
| Contact |
1-3 Tatara Miyakodani, Kyotanabe-shi, Kyoto 610-0394 Japan |
|---|
|
Faculty Molecular and Cellular Neurosciences Systems Neuroscience Brain Pathology |